Building future
worth through data
Creating future worth through data.
worth through data
Total 116 Posts
-
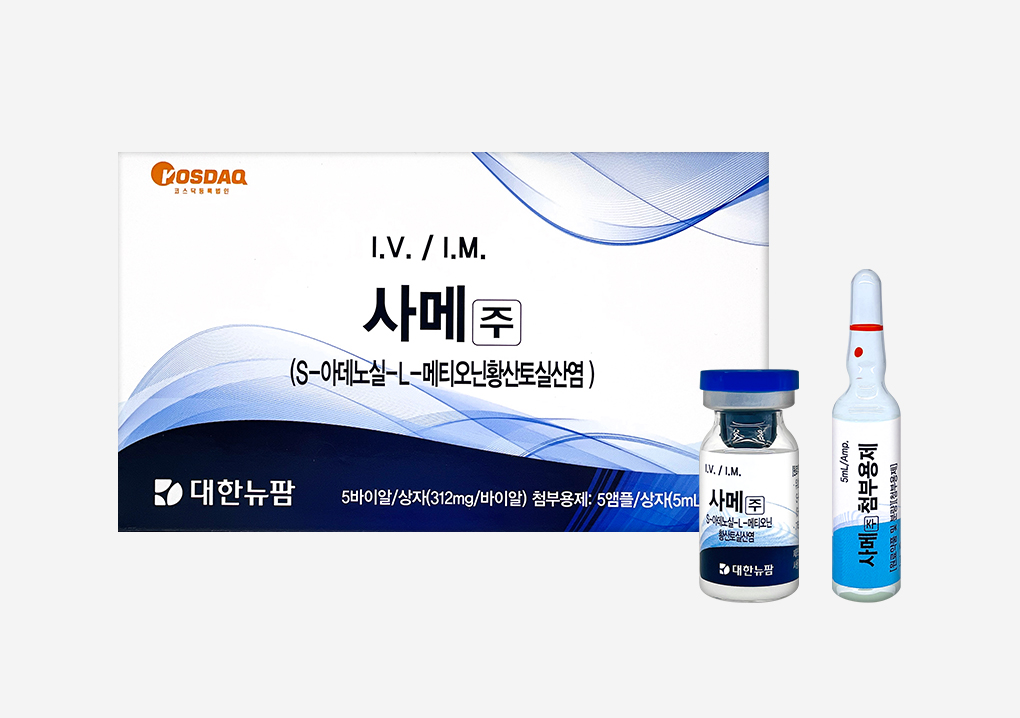 Daehan New Pharm launched the non-reimbursed injection ‘Same Injection,’ expanding its presence in the non-reimbursed injection marketDaehan New Pharm is solidifying its market position by consecutively launching non-reimbursed injections this year. Following the release of ‘Collapleo,’ a non-reimbursed injection containing atelocollagen in the first half, the company launched ‘Same injection', a non-reimbursed injection containing SAMe, on the 25th. An anticancer adjunct, ‘Thymosin Injection,’ is also scheduled for future release. ‘Same Injection' contains S-Adenosyl-L-Methionine (SAMe) as its main ingredient, which supports methylation within cells and activates homocysteine metabolism. Methylation is an epigenetic alteration affecting gene expression through chemical changes in the DNA sequence without altering the base sequence itself. Reduced methylation in cells can lead to various conditions: cancer, arteriosclerosis, and aging in adults; ADHD and autism in children; and neural tube defects in fetuses. Clinical studies have revealed that SAMe activates the TetraHydroBiopterin (BH4) cycle, which increases neurotransmitters serotonin and dopamine, improving depression, and provides strong antioxidant effects that alleviate pain from diseases such as alcoholic liver disease and arthritis. Many functional medicine and intravenous injection, experts expect ‘Same Injection', with SAMe to aid in the adjunct treatment and prevention of various diseases by acting on diverse and important metabolic mechanisms in the body. Director Ji Young-mi of the Korean Society of Intravenous Therapy (and head of the Seoul St. Mary’s Neurosurgery Functional Medicine Research Center) stated, ‘Same Injection' can provide significant help in the prevention and treatment of many intractable diseases increasing across various age groups in modern society.” ‘Same Injection' is gaining attention across diverse fields ranging from cosmetic surgery, anti-aging, wrinkle improvement, and fatigue relief to adjunctive care for cancer patients, expanding both its prescription scope and market size. According to the Korea Institute for Health and Social Affairs, the market size for non-reimbursed injections has grown rapidly every year since surpassing 1 trillion won. Alongside previously launched Iscador and Selen Injection, and upcoming Thymosin Injection, ‘Same Injection' is expected to form a key pillar in anticancer adjunctive therapy. Daehan New Pharm has ambitious plans to revamp the intravenous injection market by promoting ‘Same Injection' aggressively through online and offline channels in the second half of the year, aiming to grow it into a major pillar of their injection business.2018.06.27
Daehan New Pharm launched the non-reimbursed injection ‘Same Injection,’ expanding its presence in the non-reimbursed injection marketDaehan New Pharm is solidifying its market position by consecutively launching non-reimbursed injections this year. Following the release of ‘Collapleo,’ a non-reimbursed injection containing atelocollagen in the first half, the company launched ‘Same injection', a non-reimbursed injection containing SAMe, on the 25th. An anticancer adjunct, ‘Thymosin Injection,’ is also scheduled for future release. ‘Same Injection' contains S-Adenosyl-L-Methionine (SAMe) as its main ingredient, which supports methylation within cells and activates homocysteine metabolism. Methylation is an epigenetic alteration affecting gene expression through chemical changes in the DNA sequence without altering the base sequence itself. Reduced methylation in cells can lead to various conditions: cancer, arteriosclerosis, and aging in adults; ADHD and autism in children; and neural tube defects in fetuses. Clinical studies have revealed that SAMe activates the TetraHydroBiopterin (BH4) cycle, which increases neurotransmitters serotonin and dopamine, improving depression, and provides strong antioxidant effects that alleviate pain from diseases such as alcoholic liver disease and arthritis. Many functional medicine and intravenous injection, experts expect ‘Same Injection', with SAMe to aid in the adjunct treatment and prevention of various diseases by acting on diverse and important metabolic mechanisms in the body. Director Ji Young-mi of the Korean Society of Intravenous Therapy (and head of the Seoul St. Mary’s Neurosurgery Functional Medicine Research Center) stated, ‘Same Injection' can provide significant help in the prevention and treatment of many intractable diseases increasing across various age groups in modern society.” ‘Same Injection' is gaining attention across diverse fields ranging from cosmetic surgery, anti-aging, wrinkle improvement, and fatigue relief to adjunctive care for cancer patients, expanding both its prescription scope and market size. According to the Korea Institute for Health and Social Affairs, the market size for non-reimbursed injections has grown rapidly every year since surpassing 1 trillion won. Alongside previously launched Iscador and Selen Injection, and upcoming Thymosin Injection, ‘Same Injection' is expected to form a key pillar in anticancer adjunctive therapy. Daehan New Pharm has ambitious plans to revamp the intravenous injection market by promoting ‘Same Injection' aggressively through online and offline channels in the second half of the year, aiming to grow it into a major pillar of their injection business.2018.06.27 -
 Daehan New Pharm has filed a patent for specific fibroblast isolation, accelerating the development of skin improvement treatmentsDHNP’s research institute has registered a patent for a method to isolate and culture specific fibroblasts with superior skin regeneration effects from the heterogeneous fibroblast population, which is the main material for autologous fibroblast therapies. Existing research and products primarily isolate fibroblasts from the dermal tissue behind the patient’s ear and expand them for use in treating skin depressions, wrinkles, and refractory dermatitis. However, these therapies based on heterogeneous fibroblasts have shown lower efficacy compared to existing treatments and cosmetic fillers, leading to limited market acceptance. The research team stated, “Using this patented method, we successfully isolated papillary dermal fibroblasts, which exist in a specific dermal location, have superior proliferative ability, and play a central role in skin aging. Applying these fibroblasts in cell therapies is expected to yield significantly better effects compared to conventional fibroblast therapies.” The “High-purity Isolation Method for Papillary Dermal Fibroblasts” employs transwell technology to separate highly active papillary dermal fibroblasts from heterogeneous fibroblast populations with high purity. Cells isolated using this method demonstrated about seven times higher levels of skin regeneration factors characteristic of papillary dermal fibroblasts and exhibited excellent proliferative capacity. Furthermore, an increase in papillary dermal fibroblast factors was observed proportional to the number of culture passages, with stable maintenance and expansion confirmed. Thus, this differentiation and purification technology for specific fibroblasts establishes Daehan New Pharm’s foothold in the global cell therapy market, which is growing annually by 9.4% (expected to reach $344 billion in 2021 from $241 billion in 2017 according to Frost & Sullivan) and the domestic cell therapy market, expanding at an annual rate of 25%. Recently, DHNP relocated its bio research institute to Osong KBIO and is actively pursuing biopharmaceutical development. It has developed a high-efficiency cell culture system for fibroblast therapy and is applying serum-free media from its Japanese partner CSTI in cell therapy production.2018.05.27
Daehan New Pharm has filed a patent for specific fibroblast isolation, accelerating the development of skin improvement treatmentsDHNP’s research institute has registered a patent for a method to isolate and culture specific fibroblasts with superior skin regeneration effects from the heterogeneous fibroblast population, which is the main material for autologous fibroblast therapies. Existing research and products primarily isolate fibroblasts from the dermal tissue behind the patient’s ear and expand them for use in treating skin depressions, wrinkles, and refractory dermatitis. However, these therapies based on heterogeneous fibroblasts have shown lower efficacy compared to existing treatments and cosmetic fillers, leading to limited market acceptance. The research team stated, “Using this patented method, we successfully isolated papillary dermal fibroblasts, which exist in a specific dermal location, have superior proliferative ability, and play a central role in skin aging. Applying these fibroblasts in cell therapies is expected to yield significantly better effects compared to conventional fibroblast therapies.” The “High-purity Isolation Method for Papillary Dermal Fibroblasts” employs transwell technology to separate highly active papillary dermal fibroblasts from heterogeneous fibroblast populations with high purity. Cells isolated using this method demonstrated about seven times higher levels of skin regeneration factors characteristic of papillary dermal fibroblasts and exhibited excellent proliferative capacity. Furthermore, an increase in papillary dermal fibroblast factors was observed proportional to the number of culture passages, with stable maintenance and expansion confirmed. Thus, this differentiation and purification technology for specific fibroblasts establishes Daehan New Pharm’s foothold in the global cell therapy market, which is growing annually by 9.4% (expected to reach $344 billion in 2021 from $241 billion in 2017 according to Frost & Sullivan) and the domestic cell therapy market, expanding at an annual rate of 25%. Recently, DHNP relocated its bio research institute to Osong KBIO and is actively pursuing biopharmaceutical development. It has developed a high-efficiency cell culture system for fibroblast therapy and is applying serum-free media from its Japanese partner CSTI in cell therapy production.2018.05.27 -
 Operating profit grew significantly in the first quarter, increasing by 32% compared to the same period last yearOperating profit grew by 32% year-over-year in the first quarter. Operating profit reached 4.2 billion won, with ordinary profit at 2.2 billion won. Growth was driven by the establishment of a revenue-centered management system.2018.05.15
Operating profit grew significantly in the first quarter, increasing by 32% compared to the same period last yearOperating profit grew by 32% year-over-year in the first quarter. Operating profit reached 4.2 billion won, with ordinary profit at 2.2 billion won. Growth was driven by the establishment of a revenue-centered management system.2018.05.15 -
 Construction of an industry-academia-research collaboration center in PangyoDaehan New Pharm, together with Semyung University and the University of Suwon, has been awarded the bid for the land (E5-1 block) in Pangyo’s 2nd Techno Valley through an industry-academia-research consortium established for mutual growth. On the acquired land, an Industry-Academia-Research R&D Center with five underground floors, ten above-ground floors, and a total floor area of 9,237 m² (2,794 pyeong) is planned to be constructed, with occupancy scheduled for the end of 2020. To strengthen R&D capabilities, the consortium plans to strategically build a joint research and information exchange hub, establishing a connected system from basic research to commercialization and global market development. Furthermore, the center will provide practical training venues and opportunities for university students, open spaces for academic seminars and educational programs, and create synergy among academic, industrial, and research institutions. Specifically, within the Industry-Academia-Research Center, a Central Research Institute and Bio Research Laboratory will be established, along with Semyung University’s Clinical Trial Center for Bio-Oriental Medicine Industry and the University of Suwon’s Beauty Science Research Center, creating an integrated system for mutual growth. Research to be conducted here will include preclinical and clinical studies, skin stem cell research, development of functional oriental materials, and efficacy verification for cosmetic and pharmaceutical applications through optimized formulation R&D. The industry-academia-research collaboration will operate organically to swiftly commercialize research achievements.2018.05.04
Construction of an industry-academia-research collaboration center in PangyoDaehan New Pharm, together with Semyung University and the University of Suwon, has been awarded the bid for the land (E5-1 block) in Pangyo’s 2nd Techno Valley through an industry-academia-research consortium established for mutual growth. On the acquired land, an Industry-Academia-Research R&D Center with five underground floors, ten above-ground floors, and a total floor area of 9,237 m² (2,794 pyeong) is planned to be constructed, with occupancy scheduled for the end of 2020. To strengthen R&D capabilities, the consortium plans to strategically build a joint research and information exchange hub, establishing a connected system from basic research to commercialization and global market development. Furthermore, the center will provide practical training venues and opportunities for university students, open spaces for academic seminars and educational programs, and create synergy among academic, industrial, and research institutions. Specifically, within the Industry-Academia-Research Center, a Central Research Institute and Bio Research Laboratory will be established, along with Semyung University’s Clinical Trial Center for Bio-Oriental Medicine Industry and the University of Suwon’s Beauty Science Research Center, creating an integrated system for mutual growth. Research to be conducted here will include preclinical and clinical studies, skin stem cell research, development of functional oriental materials, and efficacy verification for cosmetic and pharmaceutical applications through optimized formulation R&D. The industry-academia-research collaboration will operate organically to swiftly commercialize research achievements.2018.05.04 -
 Launch of Specialized Therapeutic Agent for Pleuropneumonia, “Super Neofen”Daehan New Pharm has launched a new product, “Super Neofen”, developed to address swine pleuropneumonia, a contagious livestock disease. Swine pleuropneumonia, which frequently occurs during the winter season, is a bacterial infection that causes considerable damage to pig farms worldwide and has until now relied primarily on vaccination for prevention. Following years of research, “Super Neofen”, a newly developed therapeutic agent, has completed new drug registration with the Animal and Plant Quarantine Agency after passing safety and efficacy verification through domestic clinical trials. “Super Neofen” is a medication that strengthens the immune system to fight against Actinobacillus pleuropneumoniae, the causative agent of swine pleuropneumonia. It is a specialized treatment combining three active ingredients—Florfenicol, an antibiotic effective for respiratory diseases in pigs; Acetaminophen, providing antipyretic, analgesic, and anti-inflammatory effects; and Bromhexine, acting as an antitussive and expectorant. This combination delivers rapid therapeutic effects for both acute and chronic respiratory conditions. Clinical and field trials in domestic farms have shown that while the antibacterial activity of “Super Neofen” was similar to single-agent Florfenicol products, the treatment period was significantly shorter when administered compared to other formulations. The product allows simultaneous administration of the three components, making it more convenient, economical, and efficient for farmers by improving treatment outcomes, reducing labor, and enhancing productivity. Starting this month, Daehan New Pharm will begin full-scale distribution of “Super Neofen” and expand its domestic supply network aggressively. The product’s composition patent (Application No. 10-2017-0058616) has been successfully filed, and global market entry is planned for countries including the United States, China, and Russia.2017.12.20
Launch of Specialized Therapeutic Agent for Pleuropneumonia, “Super Neofen”Daehan New Pharm has launched a new product, “Super Neofen”, developed to address swine pleuropneumonia, a contagious livestock disease. Swine pleuropneumonia, which frequently occurs during the winter season, is a bacterial infection that causes considerable damage to pig farms worldwide and has until now relied primarily on vaccination for prevention. Following years of research, “Super Neofen”, a newly developed therapeutic agent, has completed new drug registration with the Animal and Plant Quarantine Agency after passing safety and efficacy verification through domestic clinical trials. “Super Neofen” is a medication that strengthens the immune system to fight against Actinobacillus pleuropneumoniae, the causative agent of swine pleuropneumonia. It is a specialized treatment combining three active ingredients—Florfenicol, an antibiotic effective for respiratory diseases in pigs; Acetaminophen, providing antipyretic, analgesic, and anti-inflammatory effects; and Bromhexine, acting as an antitussive and expectorant. This combination delivers rapid therapeutic effects for both acute and chronic respiratory conditions. Clinical and field trials in domestic farms have shown that while the antibacterial activity of “Super Neofen” was similar to single-agent Florfenicol products, the treatment period was significantly shorter when administered compared to other formulations. The product allows simultaneous administration of the three components, making it more convenient, economical, and efficient for farmers by improving treatment outcomes, reducing labor, and enhancing productivity. Starting this month, Daehan New Pharm will begin full-scale distribution of “Super Neofen” and expand its domestic supply network aggressively. The product’s composition patent (Application No. 10-2017-0058616) has been successfully filed, and global market entry is planned for countries including the United States, China, and Russia.2017.12.20 -
 Tambutin Tablet Ranked Top 3 in the Antispasmodic Category at the Russian Pharmaceutical Awards for Two Consecutive YearsTambutin Tablet (export name: Trimedat) wereed as one of the Top 3 Best Drugs in the Antispasmodic category for the second consecutive year at the Russian Pharma Awards ceremony held last month in Moscow, Russia. The Top 3ion was determined through anonymous voting by over 500,000 medical professionals registered on “Doctor at Work,” the largest professional network in Russia. The award carries special significance as the evaluation is made objectively by local healthcare specialists who assess the convenience and efficacy of pharmaceutical products. Tambutin Tablet (Trimebutine Maleate) are a convenient gastrointestinal motility regulator with various therapeutic benefits, restoring the normal physiological activity of muscles in different parts of the digestive tract. The product is on track to achieve annual sales of 5 billion KRW in Russia alone. To further enhance brand awareness, Daehan New Pharm launched a large-scale advertising campaign in October across online platforms and ten regional TV stations in Russia, including Channel One. The TV commercial, centered around the concept “smooth digestive rhythm like music,” metaphorically expresses the harmony of well-functioning digestive organs through the harmony of music, receiving highly positive feedback from local audiences. By earning continued recognition for its product excellence from both doctors and patients in Russia and maintaining active promotional efforts, the company aims to strengthen brand recognition and consumer engagement. For 2018, Daehan New Pharm has set a goal of achieving Top 3 Best Drug recognition for the third consecutive year and exceeding a 40% year-on-year sales growth target.2017.12.14
Tambutin Tablet Ranked Top 3 in the Antispasmodic Category at the Russian Pharmaceutical Awards for Two Consecutive YearsTambutin Tablet (export name: Trimedat) wereed as one of the Top 3 Best Drugs in the Antispasmodic category for the second consecutive year at the Russian Pharma Awards ceremony held last month in Moscow, Russia. The Top 3ion was determined through anonymous voting by over 500,000 medical professionals registered on “Doctor at Work,” the largest professional network in Russia. The award carries special significance as the evaluation is made objectively by local healthcare specialists who assess the convenience and efficacy of pharmaceutical products. Tambutin Tablet (Trimebutine Maleate) are a convenient gastrointestinal motility regulator with various therapeutic benefits, restoring the normal physiological activity of muscles in different parts of the digestive tract. The product is on track to achieve annual sales of 5 billion KRW in Russia alone. To further enhance brand awareness, Daehan New Pharm launched a large-scale advertising campaign in October across online platforms and ten regional TV stations in Russia, including Channel One. The TV commercial, centered around the concept “smooth digestive rhythm like music,” metaphorically expresses the harmony of well-functioning digestive organs through the harmony of music, receiving highly positive feedback from local audiences. By earning continued recognition for its product excellence from both doctors and patients in Russia and maintaining active promotional efforts, the company aims to strengthen brand recognition and consumer engagement. For 2018, Daehan New Pharm has set a goal of achieving Top 3 Best Drug recognition for the third consecutive year and exceeding a 40% year-on-year sales growth target.2017.12.14 -
 Successful Entry into the Russian Finished Pharmaceutical MarketTambutin Tablet are on track to achieve annual sales of 5 billion KRW in Russia. The export formulation, Tambutin Tablet (Trimebutine Maleate), is a gastrointestinal motility regulator known for its versatile therapeutic effects, including restoring the normal physiological activity of muscles in various parts of the digestive tract. Last year, the product was recognized as an outstanding pharmaceutical at the Russian Pharma Awards, whichs winners based on votes from Russian physicians. In Russia, Tambutin Tablet will also be featured in a nationwide TV advertising campaign starting in December—an uncommon distinction for over-the-counter medicines—indicating strong brand momentum and projecting over 40% sales growth in the coming year. Following the success of Tambutin Tablet, Daehan New Pharm has continued to achieve solid performance by completing product registrations for injectable formulations, antibiotics, and oral medications. Leveraging successful exports to Russia and its robust portfolio of wellness-oriented pharmaceuticals, the company has expanded into Central Asian markets such as Mongolia and Kazakhstan this year. From next year, Daehan New Pharm plans to strengthen its export growth further as product registrations are completed in key Southeast Asian markets, including the Philippines, Taiwan, and Vietnam.2017.11.20
Successful Entry into the Russian Finished Pharmaceutical MarketTambutin Tablet are on track to achieve annual sales of 5 billion KRW in Russia. The export formulation, Tambutin Tablet (Trimebutine Maleate), is a gastrointestinal motility regulator known for its versatile therapeutic effects, including restoring the normal physiological activity of muscles in various parts of the digestive tract. Last year, the product was recognized as an outstanding pharmaceutical at the Russian Pharma Awards, whichs winners based on votes from Russian physicians. In Russia, Tambutin Tablet will also be featured in a nationwide TV advertising campaign starting in December—an uncommon distinction for over-the-counter medicines—indicating strong brand momentum and projecting over 40% sales growth in the coming year. Following the success of Tambutin Tablet, Daehan New Pharm has continued to achieve solid performance by completing product registrations for injectable formulations, antibiotics, and oral medications. Leveraging successful exports to Russia and its robust portfolio of wellness-oriented pharmaceuticals, the company has expanded into Central Asian markets such as Mongolia and Kazakhstan this year. From next year, Daehan New Pharm plans to strengthen its export growth further as product registrations are completed in key Southeast Asian markets, including the Philippines, Taiwan, and Vietnam.2017.11.20 -
 A Milestone of Full Corporate Tax Payment and Continuous InnovationThe company announced on July 13 the results of a comprehensive corporate tax investigation through an official disclosure and completed the full payment of corporate tax amounting to 15.67 billion KRW on July 31. After completing the tax payment, the company plans to file an appeal and a petition for review in accordance with the Framework Act on National Taxes. Taking this integrated tax audit as an opportunity, Daehan New Pharm is pursuing greater internal soundness by innovating its management systems and focusing on research and development. The company’s sales grew by 7.8% in 2012 and 18.1% in 2013, maintaining an impressive annual growth rate of over 20% for three consecutive years starting in 2014. This strong growth has been driven by a well-structured product portfolio and differentiated therapeutic solutions that established Daehan New Pharm as a leader in the obesity and wellness pharmaceutical segments. In addition, continuous R&D efforts have enabled the company to expand its product lines into chronic disease treatments such as hypertension and hyperlipidemia. Since 2013, the company has also been investing aggressively in biotechnology, establishing a serum-free culture medium production facility in the Hyangnam Pharmaceutical Complex to provide customized media to clients. The company’s Central R&D Center in Hyangnam and its affiliated research institute in Osong are collaborating closely to carry out various bio-research projects. Daehan New Pharm’s focus on biotechnology stems from its strategy to combine its established strengths in obesity and wellness pharmaceuticals with the bio sector, thereby creating business synergy and new growth engines for the future. To further strengthen sales and R&D, the company continues to steadily expand its recruitment each year. Notably, all 341 employees at Daehan New Pharm are full-time regular employees—a policy aligned with the company’s management philosophy of fostering a “warm and inclusive workplace.” Every member of the organization is dedicated to sustaining the company’s strong growth trajectory and will continue to drive the momentum of innovation that has been built over the years.2017.08.02
A Milestone of Full Corporate Tax Payment and Continuous InnovationThe company announced on July 13 the results of a comprehensive corporate tax investigation through an official disclosure and completed the full payment of corporate tax amounting to 15.67 billion KRW on July 31. After completing the tax payment, the company plans to file an appeal and a petition for review in accordance with the Framework Act on National Taxes. Taking this integrated tax audit as an opportunity, Daehan New Pharm is pursuing greater internal soundness by innovating its management systems and focusing on research and development. The company’s sales grew by 7.8% in 2012 and 18.1% in 2013, maintaining an impressive annual growth rate of over 20% for three consecutive years starting in 2014. This strong growth has been driven by a well-structured product portfolio and differentiated therapeutic solutions that established Daehan New Pharm as a leader in the obesity and wellness pharmaceutical segments. In addition, continuous R&D efforts have enabled the company to expand its product lines into chronic disease treatments such as hypertension and hyperlipidemia. Since 2013, the company has also been investing aggressively in biotechnology, establishing a serum-free culture medium production facility in the Hyangnam Pharmaceutical Complex to provide customized media to clients. The company’s Central R&D Center in Hyangnam and its affiliated research institute in Osong are collaborating closely to carry out various bio-research projects. Daehan New Pharm’s focus on biotechnology stems from its strategy to combine its established strengths in obesity and wellness pharmaceuticals with the bio sector, thereby creating business synergy and new growth engines for the future. To further strengthen sales and R&D, the company continues to steadily expand its recruitment each year. Notably, all 341 employees at Daehan New Pharm are full-time regular employees—a policy aligned with the company’s management philosophy of fostering a “warm and inclusive workplace.” Every member of the organization is dedicated to sustaining the company’s strong growth trajectory and will continue to drive the momentum of innovation that has been built over the years.2017.08.02 -
 Expansion of Obesity Drug Sales – Targeting the 100-Billion-KRW Oral Obesity Treatment MarketDaehan New Pharm has recorded a 23% year-over-year growth rate in the obesity treatment market, continuing to strengthen its leadership position. The company has achieved sustained annual sales growth of over 20% for the past three years, driven by the continued launch of reimbursable products, increased market share of oral obesity drugs, and expansion into the non-reimbursable injectable treatment segment. Among these, oral obesity drugs have shown particularly steady growth. Daehan New Pharm’s representative oral anti-obesity products include appetite suppressants such as Phentin (Phendimetrazine), Phestin (Phentermine), and Phtym (Phentermine HCl), as well as the fat absorption inhibitor Zerofat (Orlistat), which the company co-markets with Mothers Pharmaceutical. In addition, Daehan New Pharm offers a wide range of obesity-related therapeutic products, collectively accounting for over 40% of the anti-obesity drug market share. Following a decline in obesity surgeries such as gastric banding and sleeve gastrectomy after the Shin Hae-chul incident, interest in combined pharmacological and exercise-based therapies has grown. As a result, the oral obesity drug market has steadily expanded, reaching approximately 100 billion KRW in 2016 according to IMS data, and continues to show growth momentum. Although newer drugs such as Belviq (Ildong Pharmaceutical) and Contrave (Kwangdong Pharmaceutical) have contributed to market expansion, traditional appetite suppressants containing phendimetrazine or phentermine and fat absorption inhibitors still hold a dominant share of the obesity market. Utilizing its extensive experience in obesity prescription marketing, Daehan New Pharm continues to expand its trading network and revenue. The company has also developed therapeutics to alleviate side effects associated with oral obesity medications, driving rapid sales growth. In the first half of this year, Daehan New Pharm’s oral obesity drug sales achieved an impressive 23% increase compared to the same period last year. Moving forward, the company plans to further strengthen brand recognition for products such as Phentin, Fastin, and Topin and expand its market share through its 3S strategy—self-control, self-confidence, and satisfaction—designed to meet the needs of both customers and patients.2017.07.25
Expansion of Obesity Drug Sales – Targeting the 100-Billion-KRW Oral Obesity Treatment MarketDaehan New Pharm has recorded a 23% year-over-year growth rate in the obesity treatment market, continuing to strengthen its leadership position. The company has achieved sustained annual sales growth of over 20% for the past three years, driven by the continued launch of reimbursable products, increased market share of oral obesity drugs, and expansion into the non-reimbursable injectable treatment segment. Among these, oral obesity drugs have shown particularly steady growth. Daehan New Pharm’s representative oral anti-obesity products include appetite suppressants such as Phentin (Phendimetrazine), Phestin (Phentermine), and Phtym (Phentermine HCl), as well as the fat absorption inhibitor Zerofat (Orlistat), which the company co-markets with Mothers Pharmaceutical. In addition, Daehan New Pharm offers a wide range of obesity-related therapeutic products, collectively accounting for over 40% of the anti-obesity drug market share. Following a decline in obesity surgeries such as gastric banding and sleeve gastrectomy after the Shin Hae-chul incident, interest in combined pharmacological and exercise-based therapies has grown. As a result, the oral obesity drug market has steadily expanded, reaching approximately 100 billion KRW in 2016 according to IMS data, and continues to show growth momentum. Although newer drugs such as Belviq (Ildong Pharmaceutical) and Contrave (Kwangdong Pharmaceutical) have contributed to market expansion, traditional appetite suppressants containing phendimetrazine or phentermine and fat absorption inhibitors still hold a dominant share of the obesity market. Utilizing its extensive experience in obesity prescription marketing, Daehan New Pharm continues to expand its trading network and revenue. The company has also developed therapeutics to alleviate side effects associated with oral obesity medications, driving rapid sales growth. In the first half of this year, Daehan New Pharm’s oral obesity drug sales achieved an impressive 23% increase compared to the same period last year. Moving forward, the company plans to further strengthen brand recognition for products such as Phentin, Fastin, and Topin and expand its market share through its 3S strategy—self-control, self-confidence, and satisfaction—designed to meet the needs of both customers and patients.2017.07.25


 Home
Home