Building future
worth through data
Creating future worth through data.
worth through data
Total 134 Posts
-
 Completion of Industry-Academia Research Center in Pangyo Second Techno ValleyDaehan New Pharm has completed the construction of its Industry-Academia Research Center in Pangyo Second Techno Valley. The newly built Daehan New Pharm Industry-Academia Research Center, located near the Daewangpangyo Interchange, spans from five basement levels to nine floors above ground and has been designed to become a new landmark in the area. The center was established with the themes of “research and development” and “communication” to enhance work efficiency and employee welfare. The basement and the first and second floors house lobbies, a main auditorium, and seminar rooms, providing spaces for collaboration. To maximize synergy among its pharmaceutical, veterinary, and bio divisions, the company relocated its Central Research Center from Hyangnam and its Bio Research Center from Osong to Pangyo. The new facility also lays the groundwork for open innovation. Daehan New Pharm will form a consortium with Semyung University’s and University of Suwon’s affiliated research institutes, which will jointly occupy the center. The company plans to continuously promote industry-academia collaboration through these partnerships. The establishment of the Daehan New Pharm Industry-Academia Research Center marks a significant milestone in the company’s growth as a pharmaceutical and biotechnology enterprise. Internally, it will serve as a workplace that fosters employee pride and engagement.2021.04.20
Completion of Industry-Academia Research Center in Pangyo Second Techno ValleyDaehan New Pharm has completed the construction of its Industry-Academia Research Center in Pangyo Second Techno Valley. The newly built Daehan New Pharm Industry-Academia Research Center, located near the Daewangpangyo Interchange, spans from five basement levels to nine floors above ground and has been designed to become a new landmark in the area. The center was established with the themes of “research and development” and “communication” to enhance work efficiency and employee welfare. The basement and the first and second floors house lobbies, a main auditorium, and seminar rooms, providing spaces for collaboration. To maximize synergy among its pharmaceutical, veterinary, and bio divisions, the company relocated its Central Research Center from Hyangnam and its Bio Research Center from Osong to Pangyo. The new facility also lays the groundwork for open innovation. Daehan New Pharm will form a consortium with Semyung University’s and University of Suwon’s affiliated research institutes, which will jointly occupy the center. The company plans to continuously promote industry-academia collaboration through these partnerships. The establishment of the Daehan New Pharm Industry-Academia Research Center marks a significant milestone in the company’s growth as a pharmaceutical and biotechnology enterprise. Internally, it will serve as a workplace that fosters employee pride and engagement.2021.04.20 -
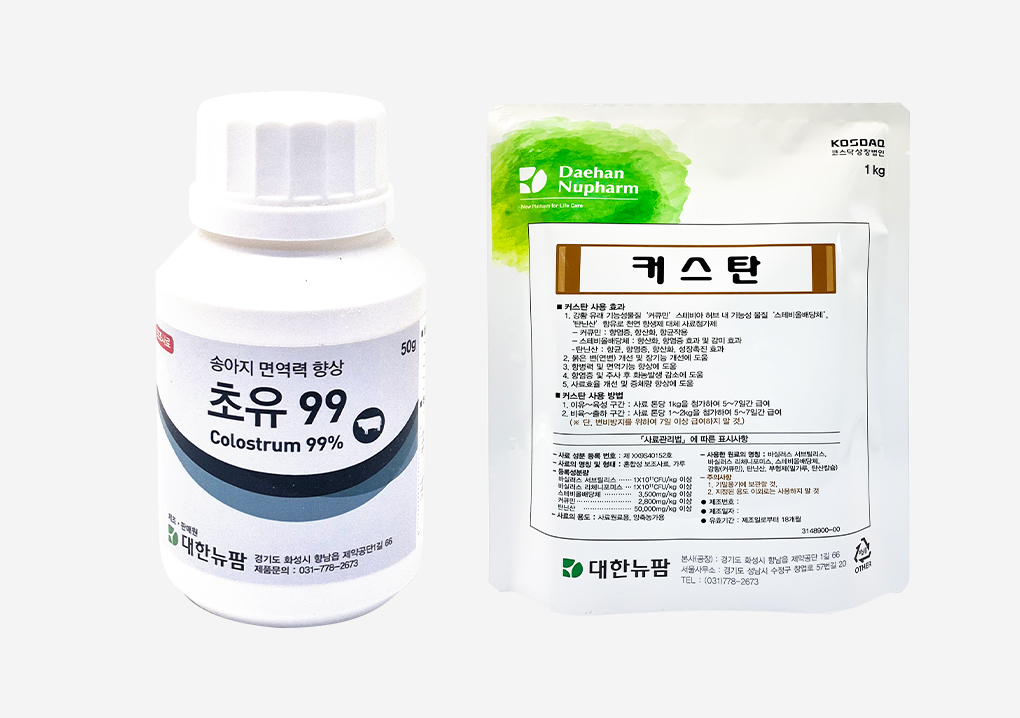 Launch of livestock disease-prevention feed supplements ‘Choyu 99’ and ‘Custan’Daehan New Pharm Co., Ltd. has launched two new livestock feed supplements, “Choyu 99” and “Custan,” designed to help prevent livestock diseases. Choyu 99 is a high-purity colostrum-based probiotic complex that promotes intestinal health in calves. The company signed a supply agreement with a leading U.S. colostrum distributor known for its premium-grade colostrum, which contains high levels of IgA (Immunoglobulin A) and IgG (Immunoglobulin G). With a purity of 99.8%, Choyu 99 provides the immune benefits of colostrum in a fast and convenient way. Colostrum feeding is recognized as the most essential factor in preventing diseases in newborn calves. Immune substances that prevent diarrhea can only be delivered through colostrum, which must be administered within 24 hours after birth. Although calves need colostrum equivalent to more than 6% of their body weight, most mother cows do not produce sufficient quantities. Therefore, feeding frozen colostrum or colostrum-based supplements has become indispensable. Custan is a natural complex formulation combining highly bioavailable curcumin with special-coated tannic acid, designed to enhance immunity and support antiviral, antibacterial, anti-inflammatory, and productivity-improving effects. Trials in growing and finishing pigs—particularly those difficult to treat during antibiotic withdrawal periods—showed that Custan significantly improved diarrhea symptoms. Moreover, when administered before and after vaccination, including foot-and-mouth disease shots, it significantly reduced abscess formation. Numerous studies and case reports have confirmed that enhanced immunity and disease resistance contribute greatly to improving productivity across various swine production stages. As a combination of these two active ingredients, Custan can deliver maximum effects even with short-term administration (5–7 days), depending on the farm’s conditions. The company stated its commitment to continued collaboration and joint development with technological partners, aiming to contribute to the livestock and related industries as a sustainable and socially responsible enterprise that pursues shared growth.2021.02.23
Launch of livestock disease-prevention feed supplements ‘Choyu 99’ and ‘Custan’Daehan New Pharm Co., Ltd. has launched two new livestock feed supplements, “Choyu 99” and “Custan,” designed to help prevent livestock diseases. Choyu 99 is a high-purity colostrum-based probiotic complex that promotes intestinal health in calves. The company signed a supply agreement with a leading U.S. colostrum distributor known for its premium-grade colostrum, which contains high levels of IgA (Immunoglobulin A) and IgG (Immunoglobulin G). With a purity of 99.8%, Choyu 99 provides the immune benefits of colostrum in a fast and convenient way. Colostrum feeding is recognized as the most essential factor in preventing diseases in newborn calves. Immune substances that prevent diarrhea can only be delivered through colostrum, which must be administered within 24 hours after birth. Although calves need colostrum equivalent to more than 6% of their body weight, most mother cows do not produce sufficient quantities. Therefore, feeding frozen colostrum or colostrum-based supplements has become indispensable. Custan is a natural complex formulation combining highly bioavailable curcumin with special-coated tannic acid, designed to enhance immunity and support antiviral, antibacterial, anti-inflammatory, and productivity-improving effects. Trials in growing and finishing pigs—particularly those difficult to treat during antibiotic withdrawal periods—showed that Custan significantly improved diarrhea symptoms. Moreover, when administered before and after vaccination, including foot-and-mouth disease shots, it significantly reduced abscess formation. Numerous studies and case reports have confirmed that enhanced immunity and disease resistance contribute greatly to improving productivity across various swine production stages. As a combination of these two active ingredients, Custan can deliver maximum effects even with short-term administration (5–7 days), depending on the farm’s conditions. The company stated its commitment to continued collaboration and joint development with technological partners, aiming to contribute to the livestock and related industries as a sustainable and socially responsible enterprise that pursues shared growth.2021.02.23 -
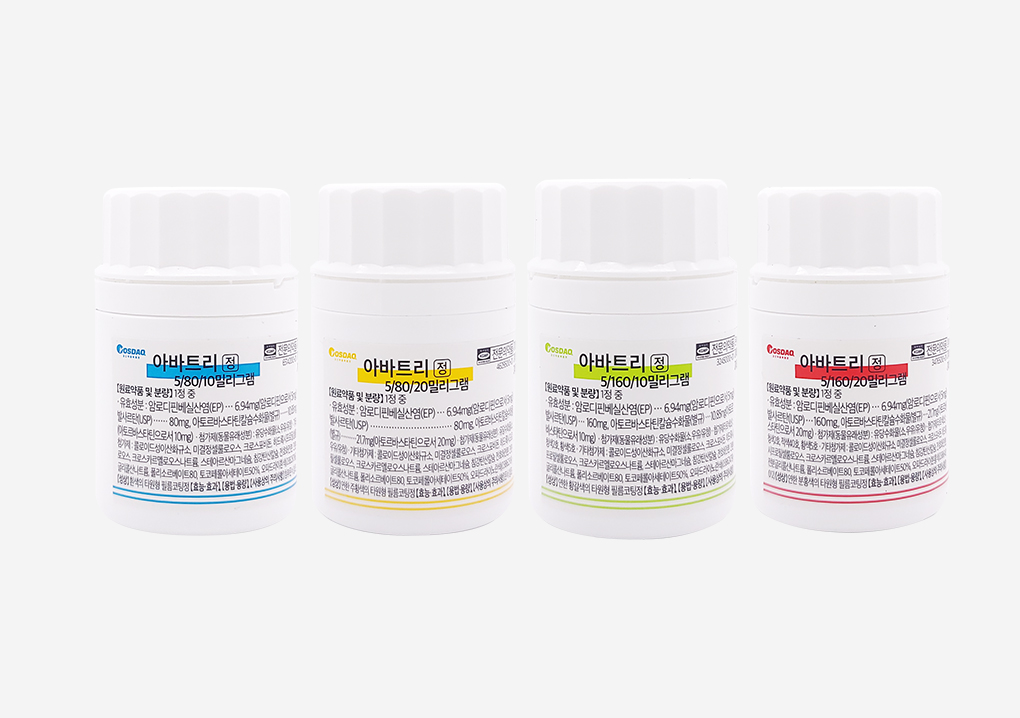 Launch of triple combination drug for hypertension and hyperlipidemia ‘Avatri Tablet’Daehan New Pharm has launched “Avatri Tablet,” the first triple combination drug in Korea containing atorvastatin, amlodipine, and valsartan. Avatri Tablet is designed to offer enhanced medication convenience for patients with hypertension and dyslipidemia. According to data from the Health Insurance Review and Assessment Service (HIRA), about 31.9% of hypertensive patients receive concurrent treatment for dyslipidemia, and the number of such comorbid cases is rapidly increasing. Combination therapies such as Avatri are known to improve long-term treatment adherence in hypertension and dyslipidemia management. Compared with patients taking separate medications, those on fixed-dose combination therapies had a 117% lower risk of discontinuation, with an average treatment duration of 35 months versus 7 months for separate dosing. Additionally, the UMPIRE study confirmed that fixed-dose combination therapy improves medication adherence by 33%. The efficacy of Avatri Tablet in controlling seated systolic blood pressure (siSBP) and low-density lipoprotein cholesterol (LDL-C) was verified through clinical trials. After eight weeks of administration, the Avatri group showed a 22.75 mmHg reduction in siSBP, compared to a 5.4 mmHg reduction in the valsartan/atorvastatin comparator group. In lipid control, Avatri also demonstrated remarkable results. After eight weeks, the Avatri -treated group showed a 47.15% reduction in LDL-C, whereas the control group receiving amlodipine/valsartan showed an increase in LDL-C levels. By combining three active ingredients to treat two diseases, Avatri provides effective regulation of lipid and blood pressure levels, making it highly beneficial for preventing cardiovascular diseases in hypertensive patients with dyslipidemia who require intensive treatment and management. The company stated that Avatri has strong potential for development in various forms, including those aimed at improving medication adherence, and reaffirmed its commitment to developing improved new drugs with superior therapeutic efficacy.2021.02.05
Launch of triple combination drug for hypertension and hyperlipidemia ‘Avatri Tablet’Daehan New Pharm has launched “Avatri Tablet,” the first triple combination drug in Korea containing atorvastatin, amlodipine, and valsartan. Avatri Tablet is designed to offer enhanced medication convenience for patients with hypertension and dyslipidemia. According to data from the Health Insurance Review and Assessment Service (HIRA), about 31.9% of hypertensive patients receive concurrent treatment for dyslipidemia, and the number of such comorbid cases is rapidly increasing. Combination therapies such as Avatri are known to improve long-term treatment adherence in hypertension and dyslipidemia management. Compared with patients taking separate medications, those on fixed-dose combination therapies had a 117% lower risk of discontinuation, with an average treatment duration of 35 months versus 7 months for separate dosing. Additionally, the UMPIRE study confirmed that fixed-dose combination therapy improves medication adherence by 33%. The efficacy of Avatri Tablet in controlling seated systolic blood pressure (siSBP) and low-density lipoprotein cholesterol (LDL-C) was verified through clinical trials. After eight weeks of administration, the Avatri group showed a 22.75 mmHg reduction in siSBP, compared to a 5.4 mmHg reduction in the valsartan/atorvastatin comparator group. In lipid control, Avatri also demonstrated remarkable results. After eight weeks, the Avatri -treated group showed a 47.15% reduction in LDL-C, whereas the control group receiving amlodipine/valsartan showed an increase in LDL-C levels. By combining three active ingredients to treat two diseases, Avatri provides effective regulation of lipid and blood pressure levels, making it highly beneficial for preventing cardiovascular diseases in hypertensive patients with dyslipidemia who require intensive treatment and management. The company stated that Avatri has strong potential for development in various forms, including those aimed at improving medication adherence, and reaffirmed its commitment to developing improved new drugs with superior therapeutic efficacy.2021.02.05 -
 Simplification of investment structureThe company had indirectly held shares of Caspian Sunrise Plc, a Kazakh oil field operator based in the United Kingdom, through Baverstock GmbH. However, as part of efforts to simplify its investment structure, the company is currently in the process of liquidating Baverstock GmbH, through which it has now come to directly hold 224,830,964 shares of Caspian Sunrise Plc.2021.01.05
Simplification of investment structureThe company had indirectly held shares of Caspian Sunrise Plc, a Kazakh oil field operator based in the United Kingdom, through Baverstock GmbH. However, as part of efforts to simplify its investment structure, the company is currently in the process of liquidating Baverstock GmbH, through which it has now come to directly hold 224,830,964 shares of Caspian Sunrise Plc.2021.01.05 -
 Patent registration for a stem cell storage excipient compositionDaehan New Pharm's Bio Research Center registered a patent on December 11 for a composition method of excipients that increase the storage period and survival rate of stem cells. Cell therapies achieve therapeutic effects by replacing damaged tissues/organs directly or through indirect actions via paracrine and endocrine factors, with ongoing research targeting various intractable diseases. Adult stem cells, especially adipose-derived mesenchymal stem cells, are advantageous due to ethical and safety benefits and ease of isolation with high cell yields. Unlike conventional drugs, cell therapies consist of living cells, resulting in a very short finished product shelf life of 1–2 days. Cell survival rates during transportation critically affect drug quality, directly limiting market competitiveness. To address these challenges, buffers to increase cell viability and rapid thawing methods post-freezing have been developed. However, toxicity and sterility issues related to commonly used DMSO freezing agents remain problematic. Daehan New Pharm developed a composition based on approved safe substances to extend storage duration and enhance cell survival rates for finished cell therapy products. Candidate substances including ascorbic acid, PDRN, pyridoxine, and DMEA wereed, and stem cell survival was evaluated over storage periods alongside analysis of post-storage stem cell characteristics. While single treatments of candidates had limited impact, a mixed composition containing ascorbic acid as the base with PDRN, pyridoxine, and DMEA significantly increased cell survival. Cell function was preserved even after more than three days of storage, as confirmed by flow cytometry and differentiation ability tests. This technology is expected to greatly extend the conventional 24-hour storage period during distribution in commercial stem cell therapies, enhancing product value and competitiveness. It is applicable not only to animal stem cells but also to human stem cells, with potential integration into various products. The Bio Research Center is dedicated to the efficient culturing, process development, transportation, and storage of stem cells, supporting the commercialization of stem cell therapies.2020.12.23
Patent registration for a stem cell storage excipient compositionDaehan New Pharm's Bio Research Center registered a patent on December 11 for a composition method of excipients that increase the storage period and survival rate of stem cells. Cell therapies achieve therapeutic effects by replacing damaged tissues/organs directly or through indirect actions via paracrine and endocrine factors, with ongoing research targeting various intractable diseases. Adult stem cells, especially adipose-derived mesenchymal stem cells, are advantageous due to ethical and safety benefits and ease of isolation with high cell yields. Unlike conventional drugs, cell therapies consist of living cells, resulting in a very short finished product shelf life of 1–2 days. Cell survival rates during transportation critically affect drug quality, directly limiting market competitiveness. To address these challenges, buffers to increase cell viability and rapid thawing methods post-freezing have been developed. However, toxicity and sterility issues related to commonly used DMSO freezing agents remain problematic. Daehan New Pharm developed a composition based on approved safe substances to extend storage duration and enhance cell survival rates for finished cell therapy products. Candidate substances including ascorbic acid, PDRN, pyridoxine, and DMEA wereed, and stem cell survival was evaluated over storage periods alongside analysis of post-storage stem cell characteristics. While single treatments of candidates had limited impact, a mixed composition containing ascorbic acid as the base with PDRN, pyridoxine, and DMEA significantly increased cell survival. Cell function was preserved even after more than three days of storage, as confirmed by flow cytometry and differentiation ability tests. This technology is expected to greatly extend the conventional 24-hour storage period during distribution in commercial stem cell therapies, enhancing product value and competitiveness. It is applicable not only to animal stem cells but also to human stem cells, with potential integration into various products. The Bio Research Center is dedicated to the efficient culturing, process development, transportation, and storage of stem cells, supporting the commercialization of stem cell therapies.2020.12.23 -
 The 3rd quarter revenue reached 113.7 billion won, marking a 15% increase year-on-yearIn 3Q 2020, Daehan New Pharm achieved a cumulative operating profit of 19.6 billion won with an operating margin of 17%, representing a 21% increase compared to the same period last year. Revenue grew 15% to 113.7 billion won, and net profit reached 6.3 billion won. The quarterly performance also showed growth momentum. Operating profit for 3Q rose 40% year-on-year to 7.4 billion won, while revenue increased 23% to 39.5 billion won. The growth was driven by a broad increase in product sales. The company launched "Fine S Tablet," a cold and allergic rhinitis medicine with bromelain as the main ingredient, and expanded sales of "Luthione Tablet," containing glutathione, well known for its use in whitening injections. Moreover, despite the challenges posed by the COVID-19 situation, the company has actively pursued research and development efforts. This overview reflects Daehan New Pharm’s strong financial performance and commitment to innovation during this period.2020.11.11
The 3rd quarter revenue reached 113.7 billion won, marking a 15% increase year-on-yearIn 3Q 2020, Daehan New Pharm achieved a cumulative operating profit of 19.6 billion won with an operating margin of 17%, representing a 21% increase compared to the same period last year. Revenue grew 15% to 113.7 billion won, and net profit reached 6.3 billion won. The quarterly performance also showed growth momentum. Operating profit for 3Q rose 40% year-on-year to 7.4 billion won, while revenue increased 23% to 39.5 billion won. The growth was driven by a broad increase in product sales. The company launched "Fine S Tablet," a cold and allergic rhinitis medicine with bromelain as the main ingredient, and expanded sales of "Luthione Tablet," containing glutathione, well known for its use in whitening injections. Moreover, despite the challenges posed by the COVID-19 situation, the company has actively pursued research and development efforts. This overview reflects Daehan New Pharm’s strong financial performance and commitment to innovation during this period.2020.11.11 -
 Daehan New Pharm achieved operating profit of 12.2 billion won in the second quarter, which is a 12.8% increase compared to the same period last yearIn the second quarter of 2020, Daehan New Pharm recorded cumulative sales of approximately 74.2 billion won, marking an 11.2% increase compared to the same period last year. Thanks to strengthened distribution management, portfolio enhancement, and consistent sales of existing key products, the company achieved an operating profit of 12.2 billion won, up 12.8% year-on-year, with an operating margin of 16.5%. Net profit also maintained a positive trend at 7.5 billion won, representing a net profit margin of 10.1%, compared to the same period last year. Steady profits were generated in the core areas of obesity and well-being pharmaceuticals, with the animal medicine business particularly driving sales growth. To sustain this momentum in the second half, the company has established detailed execution strategies by business division and is committed to strengthening its pipeline based on a new long-term vision to become a better company tomorrow.2020.08.13
Daehan New Pharm achieved operating profit of 12.2 billion won in the second quarter, which is a 12.8% increase compared to the same period last yearIn the second quarter of 2020, Daehan New Pharm recorded cumulative sales of approximately 74.2 billion won, marking an 11.2% increase compared to the same period last year. Thanks to strengthened distribution management, portfolio enhancement, and consistent sales of existing key products, the company achieved an operating profit of 12.2 billion won, up 12.8% year-on-year, with an operating margin of 16.5%. Net profit also maintained a positive trend at 7.5 billion won, representing a net profit margin of 10.1%, compared to the same period last year. Steady profits were generated in the core areas of obesity and well-being pharmaceuticals, with the animal medicine business particularly driving sales growth. To sustain this momentum in the second half, the company has established detailed execution strategies by business division and is committed to strengthening its pipeline based on a new long-term vision to become a better company tomorrow.2020.08.13 -
 Daehan New Pharm launched the external parasitic deworming product for companion animals, "Fipnil Spot-On"Daehan New Pharm Co., Ltd. (054670) has promoted the license-in of internal and external deworming products for companion animals through a strategic partnership with the Irish pharmaceutical company Chanelle Pharma Group, established in 2018. As a result, in January 2020, the company launched ‘Fipnil Spot-On,’ an animal pharmaceutical product containing the active ingredient ‘Fipronil.’ Fipronil is a phenylpyrazole compound that, when applied directly to companion animals, binds to GABA receptors in the nervous system of external parasites such as fleas and ticks. It acts as an antagonist to chloride ion channels, causing hyperpolarization that leads to parasite death. When applied to the skin of the companion animal's shoulder blade area (between both shoulders), the product spreads over the body within 24 hours through the lipid-rich layer of the skin. It concentrates and is stored in sebaceous glands and hair follicles under the skin, maintaining its effect by being released through the skin and hair for more than a month. Based on the advanced technology of Chanelle Pharma Group that maximizes the action mechanism of Fipronil, ‘Fipnil Spot-On’ has demonstrated superior product efficacy through bioequivalence tests and comparisons with original products tested not only in the EU but also in multiple countries worldwide. According to the “Survey Report on Companion Animal Ownership and Public Awareness (2017)” by the Korea Pet Food Association, among 19.56 million households in Korea, 5.63 million (28.8%) own companion animals. The estimated number of companion dogs is about 6.6 million, and companion cats about 2.07 million. Companion animal ownership has increased roughly 57% over the past five years, and this trend is expected to continue due to factors such as nuclear family structure, an increase in single-person households, aging population, and higher income levels. Additionally, the rise in vector insects due to global warming and increased contact between companion animals and wildlife vectors pose ongoing challenges. Therefore, prevention through internal and external antiparasitic products for companion animals is essential to protect both animal and owner health. The domestic market for such products has grown to over 20 billion won. The head of Daehan New Pharm's animal pharmaceuticals division stated, “By supplying the competitively positioned ‘Fipnil Spot-On’ developed through strategic cooperation with Chanelle Pharma Group’s technology, we expect to become a leading player in the steadily growing domestic internal and external deworming market for companion animals.”2020.02.05
Daehan New Pharm launched the external parasitic deworming product for companion animals, "Fipnil Spot-On"Daehan New Pharm Co., Ltd. (054670) has promoted the license-in of internal and external deworming products for companion animals through a strategic partnership with the Irish pharmaceutical company Chanelle Pharma Group, established in 2018. As a result, in January 2020, the company launched ‘Fipnil Spot-On,’ an animal pharmaceutical product containing the active ingredient ‘Fipronil.’ Fipronil is a phenylpyrazole compound that, when applied directly to companion animals, binds to GABA receptors in the nervous system of external parasites such as fleas and ticks. It acts as an antagonist to chloride ion channels, causing hyperpolarization that leads to parasite death. When applied to the skin of the companion animal's shoulder blade area (between both shoulders), the product spreads over the body within 24 hours through the lipid-rich layer of the skin. It concentrates and is stored in sebaceous glands and hair follicles under the skin, maintaining its effect by being released through the skin and hair for more than a month. Based on the advanced technology of Chanelle Pharma Group that maximizes the action mechanism of Fipronil, ‘Fipnil Spot-On’ has demonstrated superior product efficacy through bioequivalence tests and comparisons with original products tested not only in the EU but also in multiple countries worldwide. According to the “Survey Report on Companion Animal Ownership and Public Awareness (2017)” by the Korea Pet Food Association, among 19.56 million households in Korea, 5.63 million (28.8%) own companion animals. The estimated number of companion dogs is about 6.6 million, and companion cats about 2.07 million. Companion animal ownership has increased roughly 57% over the past five years, and this trend is expected to continue due to factors such as nuclear family structure, an increase in single-person households, aging population, and higher income levels. Additionally, the rise in vector insects due to global warming and increased contact between companion animals and wildlife vectors pose ongoing challenges. Therefore, prevention through internal and external antiparasitic products for companion animals is essential to protect both animal and owner health. The domestic market for such products has grown to over 20 billion won. The head of Daehan New Pharm's animal pharmaceuticals division stated, “By supplying the competitively positioned ‘Fipnil Spot-On’ developed through strategic cooperation with Chanelle Pharma Group’s technology, we expect to become a leading player in the steadily growing domestic internal and external deworming market for companion animals.”2020.02.05 -
 The product 'Vita D Injection' won the Formulation Technology Award at the 2019 International Conference of the Korean Pharmaceutical SocietyThe vitamin D injection product 'Vita D Injection' was recognized for its stability and won the Formulation Technology Award at the 2019 International Conference of the Korean Pharmaceutical Society. The award-winning technology involves suppressing the formation of soft substances (degradants) in the vitamin D injection, securing its stability, and changing the storage temperature approval from 1–25℃ to 1–30℃. This improvement increases convenience in distribution and storage. Currently, Daehan New Pharm is the only company producing vitamin D injections under the 1–30℃ storage condition. Vitamin D, specifically calciferol, is classified into D2 (ergocalciferol) and D3 (cholecalciferol). D2 is made from ergosterol, a sterol, while D3 is produced in the skin upon sunlight exposure to 7-dehydrocholesterol. Vitamin D is known for promoting calcium absorption and is part of osteoporosis treatments. Recently, its deficiency has been linked to obesity, diabetes, and heart disease. Despite the growing importance of vitamin D, modern lifestyles with reduced outdoor activity and frequent use of sunscreen make deficiency common. Since vitamin D3 easily oxidizes to form unstable soft substances, developing vitamin D injections has been challenging. Daehan New Pharm secured stabilization technology that inhibits the formation of soft substances like trans-cholecalciferol. The company registered two related patents and successfully changed storage conditions to improve convenience. The domestic market for vitamin D injections grew from 12.3 billion won in 2017 to 13.2 billion won in 2018. Daehan New Pharm produces and sells three dosage forms of 'Vita D': 100,000 IU, 200,000 IU, and 300,000 IU.2019.11.27
The product 'Vita D Injection' won the Formulation Technology Award at the 2019 International Conference of the Korean Pharmaceutical SocietyThe vitamin D injection product 'Vita D Injection' was recognized for its stability and won the Formulation Technology Award at the 2019 International Conference of the Korean Pharmaceutical Society. The award-winning technology involves suppressing the formation of soft substances (degradants) in the vitamin D injection, securing its stability, and changing the storage temperature approval from 1–25℃ to 1–30℃. This improvement increases convenience in distribution and storage. Currently, Daehan New Pharm is the only company producing vitamin D injections under the 1–30℃ storage condition. Vitamin D, specifically calciferol, is classified into D2 (ergocalciferol) and D3 (cholecalciferol). D2 is made from ergosterol, a sterol, while D3 is produced in the skin upon sunlight exposure to 7-dehydrocholesterol. Vitamin D is known for promoting calcium absorption and is part of osteoporosis treatments. Recently, its deficiency has been linked to obesity, diabetes, and heart disease. Despite the growing importance of vitamin D, modern lifestyles with reduced outdoor activity and frequent use of sunscreen make deficiency common. Since vitamin D3 easily oxidizes to form unstable soft substances, developing vitamin D injections has been challenging. Daehan New Pharm secured stabilization technology that inhibits the formation of soft substances like trans-cholecalciferol. The company registered two related patents and successfully changed storage conditions to improve convenience. The domestic market for vitamin D injections grew from 12.3 billion won in 2017 to 13.2 billion won in 2018. Daehan New Pharm produces and sells three dosage forms of 'Vita D': 100,000 IU, 200,000 IU, and 300,000 IU.2019.11.27


 Home
Home