Building future
worth through data
Creating future worth through data.
worth through data
Total 125 Posts
-
 The cumulative revenue for the third quarter reached 98.9 billion won, marking a 10% increase compared to the same period last yearIn the third quarter of 2019, Daehan New Pharm recorded cumulative sales of 98.9 billion won, representing a 10% increase compared to the same period the previous year. Operating profit rose approximately 9% year-on-year to 16.1 billion won, with an operating margin of 16.3%. Net profit also significantly increased to 9.8 billion won, reflecting a net profit margin of 9.9%. The company is divided into four main business sectors: pharmaceutical, animal pharmaceuticals, overseas business, and bio business. The pharmaceutical division, which accounts for the largest share of sales, is further categorized into reimbursed drugs, non-reimbursed drugs, and obesity and well-being injections. In 2019, sales growth was driven by new reimbursed products. Expected sales of approximately 2 billion won are projected from three products: "Hyaleum injection" for joint pain, "Newpogrel SR" effective for chronic arterial occlusion, and "NPiren S," a gastric mucosal protector. The launch of "Fine S," known for its effects on fat decomposition and sleep quality improvement, laid the foundation for growth in 2020. The company is also expanding its well-being injection field based on the immune booster "Thymosin," launched in the second half of 2018, and the functional medicine injection "Same injection," approved for depression. Despite challenges from African swine fever and livestock market downturn, the animal pharmaceuticals division grew by over 10%, overcoming a 4% decline in the domestic market. High growth was achieved through the development of differentiated and functional new products, including large-scale swine growth promoters, an improved glutathione injection for stress relief in industrial animals, and differentiated mineral blocks stimulating digestion for Korean cattle. The company plans to launch new products in the companion animal pharmaceuticals market continuously from the first half of 2020 and is working on differentiated formulation research and licensing in of European finished products. In overseas business, Daehan New Pharm expects sales growth in 2020 through active promotion and marketing of its well-being and beauty-related products, showcased at the Manila and Indonesia Philbeauty fairs. Following successful exhibitions in Southeast Asia, the company plans to set up exhibitions and exclusive booths in the emerging Latin American market in the following year to actively promote its product excellence.2019.11.12
The cumulative revenue for the third quarter reached 98.9 billion won, marking a 10% increase compared to the same period last yearIn the third quarter of 2019, Daehan New Pharm recorded cumulative sales of 98.9 billion won, representing a 10% increase compared to the same period the previous year. Operating profit rose approximately 9% year-on-year to 16.1 billion won, with an operating margin of 16.3%. Net profit also significantly increased to 9.8 billion won, reflecting a net profit margin of 9.9%. The company is divided into four main business sectors: pharmaceutical, animal pharmaceuticals, overseas business, and bio business. The pharmaceutical division, which accounts for the largest share of sales, is further categorized into reimbursed drugs, non-reimbursed drugs, and obesity and well-being injections. In 2019, sales growth was driven by new reimbursed products. Expected sales of approximately 2 billion won are projected from three products: "Hyaleum injection" for joint pain, "Newpogrel SR" effective for chronic arterial occlusion, and "NPiren S," a gastric mucosal protector. The launch of "Fine S," known for its effects on fat decomposition and sleep quality improvement, laid the foundation for growth in 2020. The company is also expanding its well-being injection field based on the immune booster "Thymosin," launched in the second half of 2018, and the functional medicine injection "Same injection," approved for depression. Despite challenges from African swine fever and livestock market downturn, the animal pharmaceuticals division grew by over 10%, overcoming a 4% decline in the domestic market. High growth was achieved through the development of differentiated and functional new products, including large-scale swine growth promoters, an improved glutathione injection for stress relief in industrial animals, and differentiated mineral blocks stimulating digestion for Korean cattle. The company plans to launch new products in the companion animal pharmaceuticals market continuously from the first half of 2020 and is working on differentiated formulation research and licensing in of European finished products. In overseas business, Daehan New Pharm expects sales growth in 2020 through active promotion and marketing of its well-being and beauty-related products, showcased at the Manila and Indonesia Philbeauty fairs. Following successful exhibitions in Southeast Asia, the company plans to set up exhibitions and exclusive booths in the emerging Latin American market in the following year to actively promote its product excellence.2019.11.12 -
 Daehan New Pharm achieved sales of 66.8 billion won and operating profit of 10.8 billion won in the first half of the yearIn the first half of 2019, Daehan New Pharm achieved sales of 66.8 billion won, marking an 8.6% increase compared to the same period the previous year. Operating profit rose 14.5% to 10.8 billion won, and net profit turned positive, reaching 6.6 billion won. Overall performance improved due to company-wide enhancements in sales efficiency and increased revenue from new product launches, while steady sales continued for existing key products. Steady profits were also generated in the core sectors of obesity and well-being pharmaceuticals, with strong sales expected in the second half of the year. The company plans to maximize performance through visionary management strategies and strengthening its new product portfolio. Financial structure improvements initiated last year and favorable reception of new products contributed to the improved results. The company aims to maintain operational efficiency and continue launching new products to sustain positive momentum throughout the second half.2019.08.12
Daehan New Pharm achieved sales of 66.8 billion won and operating profit of 10.8 billion won in the first half of the yearIn the first half of 2019, Daehan New Pharm achieved sales of 66.8 billion won, marking an 8.6% increase compared to the same period the previous year. Operating profit rose 14.5% to 10.8 billion won, and net profit turned positive, reaching 6.6 billion won. Overall performance improved due to company-wide enhancements in sales efficiency and increased revenue from new product launches, while steady sales continued for existing key products. Steady profits were also generated in the core sectors of obesity and well-being pharmaceuticals, with strong sales expected in the second half of the year. The company plans to maximize performance through visionary management strategies and strengthening its new product portfolio. Financial structure improvements initiated last year and favorable reception of new products contributed to the improved results. The company aims to maintain operational efficiency and continue launching new products to sustain positive momentum throughout the second half.2019.08.12 -
 Daehan New Pharm has filed a patent for a method of specifically isolating fibroblasts, accelerating the development of skin improvement treatmentsDaehan New Pharm registered a patent for a method to specifically isolate heterogenetic fibroblasts with excellent skin regeneration effects by separating and culturing only those fibroblasts. Traditional therapies use fibroblasts primarily isolated from dermal tissue behind the ear for treating skin depressions, wrinkles, and refractory dermatitis. However, these treatments are based on mixed fibroblasts, resulting in lower efficacy compared to targeted therapies and cosmetic fillers. The patented method successfully isolates papillary dermal fibroblasts, located in specific dermal areas, known for high proliferative capacity and a central role in skin aging. Applying these in cell therapies is expected to show superior effects compared to conventional fibroblast treatments. The “High-purity isolation method for papillary dermal fibroblasts” uses transwell technology to separate highly active papillary dermal fibroblasts from mixed fibroblast populations. Cells isolated with this method showed about seven times higher levels of papillary dermal fibroblast factors and superior proliferative ability. This increase correlates with repeated culturing and maintaining processes. This purification technology for specific fibroblasts marks Daehan New Pharm's full-scale entry into the cell therapy market. According to Frost & Sullivan, the global cell therapy market is projected to grow at a CAGR of 9.4%, reaching $344 billion by 2021, while the domestic market is growing annually by 25%. Recently, the Bio Research Center relocated to Osong KBIO and is actively advancing biopharmaceutical development. They have developed a high-efficiency cell culture system for fibroblast therapies and are applying serum-free media from their Japanese partner CSTI in cell therapy development.2019.05.28
Daehan New Pharm has filed a patent for a method of specifically isolating fibroblasts, accelerating the development of skin improvement treatmentsDaehan New Pharm registered a patent for a method to specifically isolate heterogenetic fibroblasts with excellent skin regeneration effects by separating and culturing only those fibroblasts. Traditional therapies use fibroblasts primarily isolated from dermal tissue behind the ear for treating skin depressions, wrinkles, and refractory dermatitis. However, these treatments are based on mixed fibroblasts, resulting in lower efficacy compared to targeted therapies and cosmetic fillers. The patented method successfully isolates papillary dermal fibroblasts, located in specific dermal areas, known for high proliferative capacity and a central role in skin aging. Applying these in cell therapies is expected to show superior effects compared to conventional fibroblast treatments. The “High-purity isolation method for papillary dermal fibroblasts” uses transwell technology to separate highly active papillary dermal fibroblasts from mixed fibroblast populations. Cells isolated with this method showed about seven times higher levels of papillary dermal fibroblast factors and superior proliferative ability. This increase correlates with repeated culturing and maintaining processes. This purification technology for specific fibroblasts marks Daehan New Pharm's full-scale entry into the cell therapy market. According to Frost & Sullivan, the global cell therapy market is projected to grow at a CAGR of 9.4%, reaching $344 billion by 2021, while the domestic market is growing annually by 25%. Recently, the Bio Research Center relocated to Osong KBIO and is actively advancing biopharmaceutical development. They have developed a high-efficiency cell culture system for fibroblast therapies and are applying serum-free media from their Japanese partner CSTI in cell therapy development.2019.05.28 -
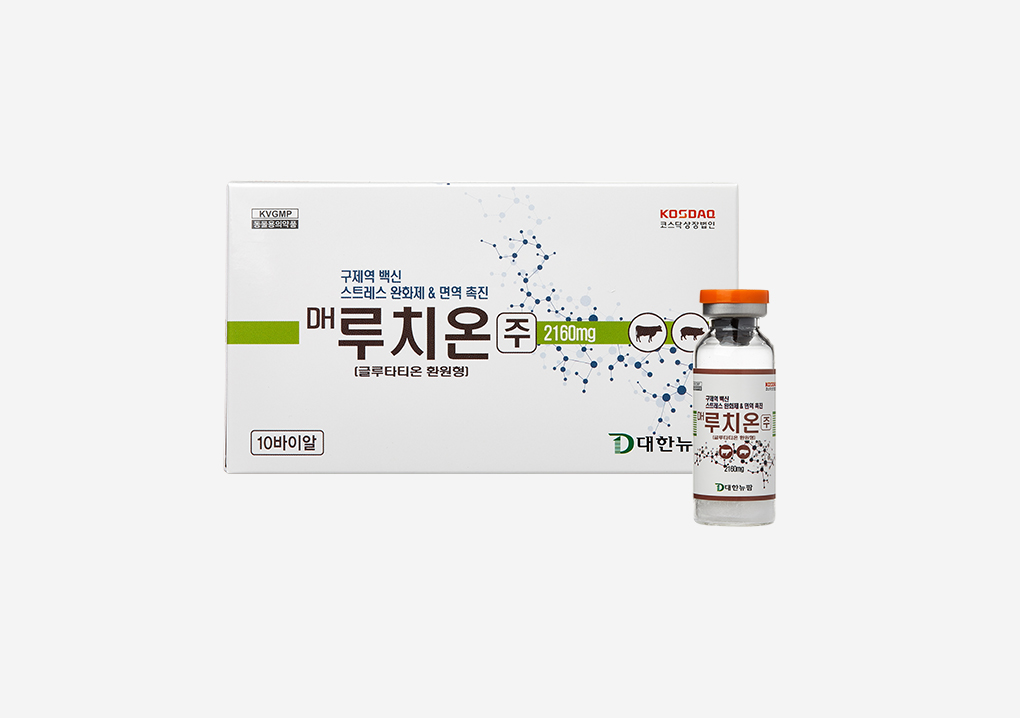 Daehan New Pharm has launched a new drug designed to relieve stress and boost immunity for foot-and-mouth disease vaccinesDaehan New Pharm launched ‘DH Luthione Injection,’ a glutathione (GSH, glutathione sulfhydryl) drug recognized for its powerful antioxidant function, detoxification, and immune-enhancing effects. Glutathione is a small protein composed of three amino acids—cysteine, glutamic acid, and glycine—produced mainly in animal cells, especially in the liver. Its primary functions include antioxidation, detoxification, immune system enhancement, and anti-inflammatory effects. Other antioxidants such as vitamins C and E depend on glutathione, making it the most potent antioxidant known. In industrial animals, glutathione’s functions can prevent productivity loss caused by various stresses and frequent drug exposure, supporting its long-term use in livestock. However, most glutathione products approved for animals are oral formulations, which show low bioavailability due to poor absorption, limiting their effectiveness. Particularly during vaccinations such as foot-and-mouth disease, there was a need to develop injectable formulations that maximize stress relief, reduce inflammation, and boost immunity. To maximize effects, Daehan New Pharm used the same formulation and high-quality raw materials (from Kyowa Hakko Bio Co., Ltd.) as its leading human glutathione injection ‘Luthione Injection’ to develop ‘DH Luthione,’ the first freeze-dried glutathione injectable for animals. Clinical trials at Gyeongsang National University Veterinary College demonstrated that ‘DH Luthione’ effectively reduced stress and inflammation markers (cortisol, TNF-α, CRP) after foot-and-mouth disease vaccination. It significantly increased cell-mediated immunity (IFN-γ) and humoral immunity markers (IgG, IgM, IgA), and improved liver function markers (AST, ALT, bilirubin) while raising albumin levels. These verified benefits make ‘DH Luthione’ a valuable agent for enhancing productivity in industrial animals.2019.03.20
Daehan New Pharm has launched a new drug designed to relieve stress and boost immunity for foot-and-mouth disease vaccinesDaehan New Pharm launched ‘DH Luthione Injection,’ a glutathione (GSH, glutathione sulfhydryl) drug recognized for its powerful antioxidant function, detoxification, and immune-enhancing effects. Glutathione is a small protein composed of three amino acids—cysteine, glutamic acid, and glycine—produced mainly in animal cells, especially in the liver. Its primary functions include antioxidation, detoxification, immune system enhancement, and anti-inflammatory effects. Other antioxidants such as vitamins C and E depend on glutathione, making it the most potent antioxidant known. In industrial animals, glutathione’s functions can prevent productivity loss caused by various stresses and frequent drug exposure, supporting its long-term use in livestock. However, most glutathione products approved for animals are oral formulations, which show low bioavailability due to poor absorption, limiting their effectiveness. Particularly during vaccinations such as foot-and-mouth disease, there was a need to develop injectable formulations that maximize stress relief, reduce inflammation, and boost immunity. To maximize effects, Daehan New Pharm used the same formulation and high-quality raw materials (from Kyowa Hakko Bio Co., Ltd.) as its leading human glutathione injection ‘Luthione Injection’ to develop ‘DH Luthione,’ the first freeze-dried glutathione injectable for animals. Clinical trials at Gyeongsang National University Veterinary College demonstrated that ‘DH Luthione’ effectively reduced stress and inflammation markers (cortisol, TNF-α, CRP) after foot-and-mouth disease vaccination. It significantly increased cell-mediated immunity (IFN-γ) and humoral immunity markers (IgG, IgM, IgA), and improved liver function markers (AST, ALT, bilirubin) while raising albumin levels. These verified benefits make ‘DH Luthione’ a valuable agent for enhancing productivity in industrial animals.2019.03.20 -
 Daehan New Pharm has officially entered the facial thread lifting market with the launch of ‘TASS-UP,’ a compressed cog thread product designed for facial liftingDaehan New Pharm launched ‘TASS-UP,’ a compressed cog thread product for the facial thread lifting market. In August 2018, the company signed an exclusive sales agreement with Gishell Group (CEO Kim Jong-woo) for the premium facial thread lift product ‘TESSLIFTSOFT.’ Following this, by releasing TASS-UP, which is the most popularly used compressed cog thread in the market, Daehan New Pharm has further strengthened its position in the facial thread lifting sector. Facial thread lifting, one of the most popular procedures for maintaining a youthful face, has recently gained popularity over methods like Botox, which have short durations, or fillers that may carry higher risks of side effects. TESSLIFT, launched last year, is known for its 3D mesh structure that provides natural lifting and volumizing effects, with a longer-lasting impact that has made it especially popular among young women. However, patients with sagging skin or deep nasolabial folds require stronger lifting effects. Industry experts emphasize that a combination treatment of compressed cog threads like TASS-UP is necessary for such cases. TASS-UP features thicker cogs than traditional sliced-cut threads, resulting in longer-lasting and more powerful lifting effects, making it suitable for a variety of patients. It is produced using a non-heating process, minimizing degradation of thread components due to heat, which addresses the common problem of thread breakage during long-term storage. Unlike bidirectional cogs, TASS-UP’s three-dimensional cogs are twisted at a consistent angle, enhancing lifting effectiveness. According to ISAPS (International Society of Aesthetic and Plastic Surgery), Korea’s face lifting market is expected to surge from approximately 10 billion won in 2016 to about 35 billion won by 2020. Thread lifting procedures are favored by clinicians due to their relative ease of performance and low incidence of side effects. Dr. Choi Ho-seong of Pionacl Clinic stated, “By using the newly released TASS-UP to achieve strong lifting effects and appropriately combining it with the natural, firm, and long-lasting TESSLIFTSOFT, patient satisfaction can be greatly increased.” Dr. Lee Sang-bong of Wonjin Dermatology Clinic noted, “TASS-UP and TESSLIFTSOFT not only produce synergistic effects but also complement the shortcomings of each thread, enhancing both efficacy and cost-effectiveness.” Daehan New Pharm is strengthening its position as a pharmaceutical bio company specializing in obesity and beauty products. Following a stable launch of facial thread lifting, the company plans to expand its portfolio of facial aesthetic products by introducing various medical devices.2019.03.05
Daehan New Pharm has officially entered the facial thread lifting market with the launch of ‘TASS-UP,’ a compressed cog thread product designed for facial liftingDaehan New Pharm launched ‘TASS-UP,’ a compressed cog thread product for the facial thread lifting market. In August 2018, the company signed an exclusive sales agreement with Gishell Group (CEO Kim Jong-woo) for the premium facial thread lift product ‘TESSLIFTSOFT.’ Following this, by releasing TASS-UP, which is the most popularly used compressed cog thread in the market, Daehan New Pharm has further strengthened its position in the facial thread lifting sector. Facial thread lifting, one of the most popular procedures for maintaining a youthful face, has recently gained popularity over methods like Botox, which have short durations, or fillers that may carry higher risks of side effects. TESSLIFT, launched last year, is known for its 3D mesh structure that provides natural lifting and volumizing effects, with a longer-lasting impact that has made it especially popular among young women. However, patients with sagging skin or deep nasolabial folds require stronger lifting effects. Industry experts emphasize that a combination treatment of compressed cog threads like TASS-UP is necessary for such cases. TASS-UP features thicker cogs than traditional sliced-cut threads, resulting in longer-lasting and more powerful lifting effects, making it suitable for a variety of patients. It is produced using a non-heating process, minimizing degradation of thread components due to heat, which addresses the common problem of thread breakage during long-term storage. Unlike bidirectional cogs, TASS-UP’s three-dimensional cogs are twisted at a consistent angle, enhancing lifting effectiveness. According to ISAPS (International Society of Aesthetic and Plastic Surgery), Korea’s face lifting market is expected to surge from approximately 10 billion won in 2016 to about 35 billion won by 2020. Thread lifting procedures are favored by clinicians due to their relative ease of performance and low incidence of side effects. Dr. Choi Ho-seong of Pionacl Clinic stated, “By using the newly released TASS-UP to achieve strong lifting effects and appropriately combining it with the natural, firm, and long-lasting TESSLIFTSOFT, patient satisfaction can be greatly increased.” Dr. Lee Sang-bong of Wonjin Dermatology Clinic noted, “TASS-UP and TESSLIFTSOFT not only produce synergistic effects but also complement the shortcomings of each thread, enhancing both efficacy and cost-effectiveness.” Daehan New Pharm is strengthening its position as a pharmaceutical bio company specializing in obesity and beauty products. Following a stable launch of facial thread lifting, the company plans to expand its portfolio of facial aesthetic products by introducing various medical devices.2019.03.05 -
 Daehan New Pharm achieved a cumulative operating profit of 14.8 billion won in the third quarter, representing a 44.5% increase compared to the same period last yearDaehan New Pharm announced on the 14th that its cumulative operating profit for the third quarter increased approximately 44.5% year-on-year to 14.8 billion won. Sales declined about 9% to 90.2 billion won, while net profit turned positive. The significant improvement was driven by expanded sales of high-margin products and the establishment of an efficient management system. The company has been strengthening its product portfolio, improving distribution channels, and optimizing costs, creating a virtuous cycle by reinvesting profits into research and development to drive future growth. Operating efficiency enhancements contributed to performance improvements, with strategic new products such as ‘Same injection’ ‘Thymosin injection,’ and ‘TESSLIFTSOFT’ at the core of ongoing growth efforts. ‘Same injection,’ approved as a treatment for depression, is also known for its excellent effects in cancer adjunct therapy, liver function improvement, and pain management, and is expected to continue the reputation of ‘Luthione Injection’ (commonly known as ‘Baekok Injection’) as a sales leader. The company has an exclusive sales agreement for the premium mesh-structured dissolvable thread ‘TESSLIFTSOFT’ and is actively marketing its differentiated product strengths. Through diversification of sales and improvement of profitability structure, sales increased from 56.7 billion won in 2013 to 130.8 billion won last year, while operating profit rose from 6 billion won to 13.1 billion won.2018.11.14
Daehan New Pharm achieved a cumulative operating profit of 14.8 billion won in the third quarter, representing a 44.5% increase compared to the same period last yearDaehan New Pharm announced on the 14th that its cumulative operating profit for the third quarter increased approximately 44.5% year-on-year to 14.8 billion won. Sales declined about 9% to 90.2 billion won, while net profit turned positive. The significant improvement was driven by expanded sales of high-margin products and the establishment of an efficient management system. The company has been strengthening its product portfolio, improving distribution channels, and optimizing costs, creating a virtuous cycle by reinvesting profits into research and development to drive future growth. Operating efficiency enhancements contributed to performance improvements, with strategic new products such as ‘Same injection’ ‘Thymosin injection,’ and ‘TESSLIFTSOFT’ at the core of ongoing growth efforts. ‘Same injection,’ approved as a treatment for depression, is also known for its excellent effects in cancer adjunct therapy, liver function improvement, and pain management, and is expected to continue the reputation of ‘Luthione Injection’ (commonly known as ‘Baekok Injection’) as a sales leader. The company has an exclusive sales agreement for the premium mesh-structured dissolvable thread ‘TESSLIFTSOFT’ and is actively marketing its differentiated product strengths. Through diversification of sales and improvement of profitability structure, sales increased from 56.7 billion won in 2013 to 130.8 billion won last year, while operating profit rose from 6 billion won to 13.1 billion won.2018.11.14 -
 Daehan New Pharm achieved an operating profit of 9.4 billion won in the first half of the year, representing a 44% increase compared to the same period last yearIn the first half of 2018, Daehan New Pharm recorded sales of 61.469 billion won, a decrease of approximately 5% compared to the same period last year. However, operating profit rose significantly by about 44% to 9.46 billion won from 6.563 billion won in the previous year. Due to increased other expenses, the company recorded a net loss of 3.881 billion won. Gradual improvements in financial structure and cost efficiency have steadily strengthened the company's fundamentals. The newly launched non-reimbursed injection ‘Same injection’ and the upcoming anticancer adjunct ‘Thymosin injection’ are expected to contribute to sales growth in the second half, supported by active seminars and both online and offline promotions.2018.08.14
Daehan New Pharm achieved an operating profit of 9.4 billion won in the first half of the year, representing a 44% increase compared to the same period last yearIn the first half of 2018, Daehan New Pharm recorded sales of 61.469 billion won, a decrease of approximately 5% compared to the same period last year. However, operating profit rose significantly by about 44% to 9.46 billion won from 6.563 billion won in the previous year. Due to increased other expenses, the company recorded a net loss of 3.881 billion won. Gradual improvements in financial structure and cost efficiency have steadily strengthened the company's fundamentals. The newly launched non-reimbursed injection ‘Same injection’ and the upcoming anticancer adjunct ‘Thymosin injection’ are expected to contribute to sales growth in the second half, supported by active seminars and both online and offline promotions.2018.08.14 -
 Operating profit increased significantly / Strong sales of new products are expected in the second half of the year (based on the half-year financial disclosure)Daehan New Pharm announced that its consolidated operating profit for the first half of 2018 reached 9.46 billion won, representing a 44% increase compared to the same period the previous year. Sales slightly decreased to 61.469 billion won, and net loss amounted to 3.881 billion won due to increased other expenses. The company has been strengthening its market position recently with the launch of the non-reimbursed injection ‘Same injection’ and plans to release the anticancer adjunct ‘Thymosin Injection.’ In July, Daehan New Pharm signed an exclusive sales contract for the next-generation premium dissolvable thread ‘TESSLIFTSOFT.’ Demand for the livestock stress reliever ‘Power GABA-C’ surged due to the record heatwave. Power GABA-C is user-friendly and proven effective against stress, dehydration, and reproductive disorders, with demand already exceeding production plans. Continuous efforts to improve financial structure and optimize costs are showing positive results. The new injection ‘Same injection’ and the upcoming anticancer adjunct ‘Thymosin Injection’ are expected to contribute to sales growth in the second half, supported by active seminars and online/offline promotions.2018.08.14
Operating profit increased significantly / Strong sales of new products are expected in the second half of the year (based on the half-year financial disclosure)Daehan New Pharm announced that its consolidated operating profit for the first half of 2018 reached 9.46 billion won, representing a 44% increase compared to the same period the previous year. Sales slightly decreased to 61.469 billion won, and net loss amounted to 3.881 billion won due to increased other expenses. The company has been strengthening its market position recently with the launch of the non-reimbursed injection ‘Same injection’ and plans to release the anticancer adjunct ‘Thymosin Injection.’ In July, Daehan New Pharm signed an exclusive sales contract for the next-generation premium dissolvable thread ‘TESSLIFTSOFT.’ Demand for the livestock stress reliever ‘Power GABA-C’ surged due to the record heatwave. Power GABA-C is user-friendly and proven effective against stress, dehydration, and reproductive disorders, with demand already exceeding production plans. Continuous efforts to improve financial structure and optimize costs are showing positive results. The new injection ‘Same injection’ and the upcoming anticancer adjunct ‘Thymosin Injection’ are expected to contribute to sales growth in the second half, supported by active seminars and online/offline promotions.2018.08.14 -
 Daehan New Pharm launched 'DH Pulvisin Granules,' a treatment for skin fungal infections in dogs and cats'DH Pulvisin Granules' was launched in June as a prevention and treatment for skin fungal infections caused by fungi such as Tinea, Trichophyton, and Microsporum, which are major skin diseases in companion animals (dogs and cats). Skin fungal infections in companion animals, known as ringworm, are highly contagious zoonotic diseases that can easily spread to owners and cohabiting pets if not properly treated. Various antifungal agents are used for treatment. The active ingredient in 'DH Pulvisin Granules', griseofulvin, is widely known to be highly effective against skin fungal infections. However, the use of human drugs without confirmed efficacy or safety in companion animals has been common. Clinical and toxicity trials of 'DH Pulvisin Granules' conducted at Seoul National University Veterinary College confirmed its excellent efficacy and safety in treating skin fungal infections in companion animals. It received approval in February 2018 as the only veterinary drug containing griseofulvin. This is not merely a substitute for human drugs but the only product scientifically and systematically verified in animals, offering significant assistance in treating skin fungal infections in companion animals.2018.07.02
Daehan New Pharm launched 'DH Pulvisin Granules,' a treatment for skin fungal infections in dogs and cats'DH Pulvisin Granules' was launched in June as a prevention and treatment for skin fungal infections caused by fungi such as Tinea, Trichophyton, and Microsporum, which are major skin diseases in companion animals (dogs and cats). Skin fungal infections in companion animals, known as ringworm, are highly contagious zoonotic diseases that can easily spread to owners and cohabiting pets if not properly treated. Various antifungal agents are used for treatment. The active ingredient in 'DH Pulvisin Granules', griseofulvin, is widely known to be highly effective against skin fungal infections. However, the use of human drugs without confirmed efficacy or safety in companion animals has been common. Clinical and toxicity trials of 'DH Pulvisin Granules' conducted at Seoul National University Veterinary College confirmed its excellent efficacy and safety in treating skin fungal infections in companion animals. It received approval in February 2018 as the only veterinary drug containing griseofulvin. This is not merely a substitute for human drugs but the only product scientifically and systematically verified in animals, offering significant assistance in treating skin fungal infections in companion animals.2018.07.02


 Home
Home